  The entropy of a substance is zero if the absolute temperature is zero. Common thermodynamic terms and functions - potential energy, kinetic energy, thermal or internal energy, chemical energy, nuclear energy and more. For the d-dimensional case we devise a Monte Carlo method based upon the properties of subadditive processes and superconvolutive sequences. especially the relation between mass and luminosity is very tight. Thermodynamic Terms - Functions and Relations. We obtain an analytic solution of this example in the 1-dimensional case through an exploitation of the theory of regenerative phenomena. This general theory is illustrated by an example, in which the potential is proportional to the volume covered by interpenetrating unit spheres centred at the particles. The relation turns out to be a certain involution on the space of functions of two variables. 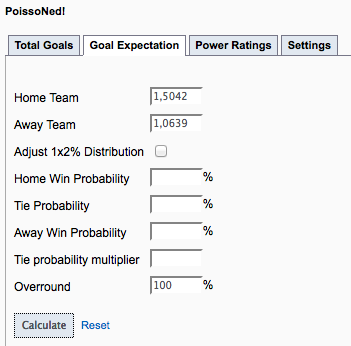
We study the cumulant functions of an arbitrary function (e.g., a potential) of the position of the particles we find the relationship between the two respective cumulant functions and we examine how they depend upon the thermodynamic partition functions. 16, 199–206 (1988).The uniform random distribution of particles in a region of space may follow either of two models, multinomial or Poisson, according as the number of particles in the region is respectively fixed or random. Angenheister, G.) 33 (Springer, Berlin, 1982). #Poisson relations thermodynamics calculator series#Landolt-Börnstein: Numerical Data and Functional Relationships in Science and Technology, new series V/1b (ed. Thermophysical Properties of Materials, 62 (North-Holland, Amsterdam, 1986). calculation: for example, whereas the relationship between energy, heat. Being an inexact differential, dW is sometimes denoted as W (Tsonis) or DW (notes below). Substituting this in the above yields or where the kinetic energy K mv2. What is the specific volume of the parcel d. How many moles of air are in the parcel (The molecular weight of air is 28.96 g/mol) b. A 1.5-kg parcel of dry air is at a temperature of 15 C and a pressure of 1013 mb. A Treatise on the Mathematical Theory of Elasticity, 4th Edn 163 (Dover, New York, 1944). Thermodynamics of Open Systems and the Meaning of Chemical Potential116. Since p F/A (or F pA), the above equation becomes dW Fvdt or dW/dt Fv Now from Newton’s Law, F ma mdv/dt. Starting with cpdT dq +dp, derive the Poisson relation T p(1)const. We attribute the occurrence of a negative Poisson ratio in low-density silica polymorphs to the high rigidity of the SiO 4 tetrahedra. Hookes law, Youngs modulus, bulk modulus, shear, modulus of rigidity, Poissons ratio elastic energy. This is the first of many thermodynamic relations, generally called Maxwell. Elastic behaviour, stress-strain relationship. In the present paper we calculate the elastic properties of the hard-sphere. Our calculations reproduce the negative Poisson ratio in α-cristobalite, and predict that α-quartz, the most common form of crystalline silica, will also exhibit a negative Poisson ratio under large uniaxial tension. Given a probability distribution, we can calculate the expected value of any. Elastic constants and the Poisson ratio of the fcc hard-sphere crystalline. We have investigated the elastic behaviour of α-cristobalite and other forms of silica with first-principles calculations and classical interatomic potentials. 
Although a negative Poisson ratio is not forbidden by thermodynamics, it is rare in crystalline solids: the results of recent experiments 1 which observed a negative Poisson ratio in α-cristobalite were therefore unexpected. Normally, this ratio is positive, as most solids expand in the transverse direction when subjected to a uniaxial compression. 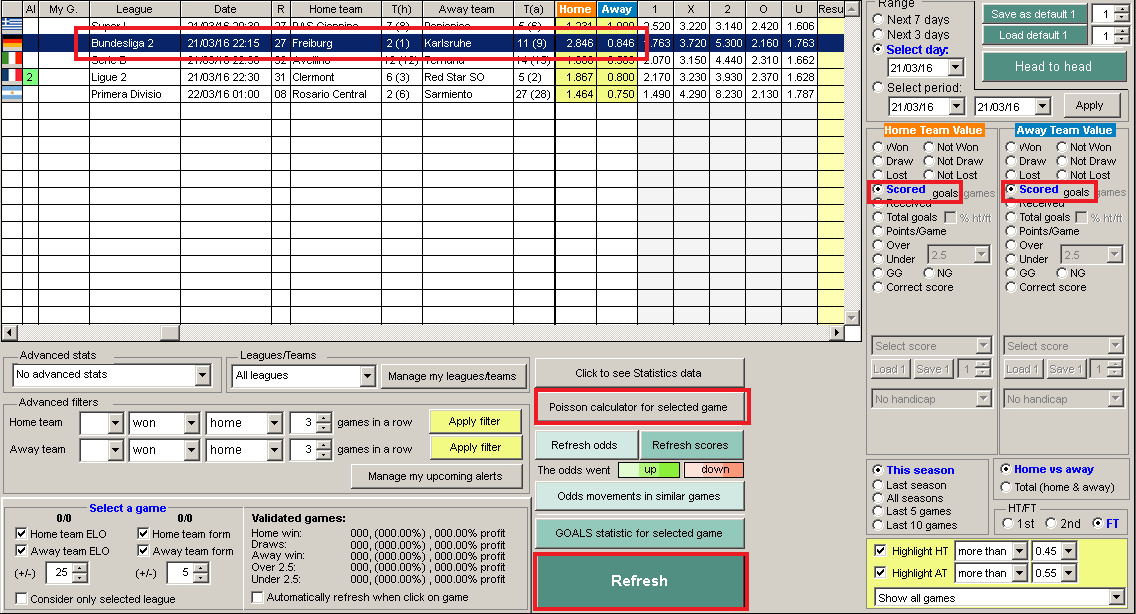
enters the calculation inverse in the thermodynamic ( 1. It is defined as the negative ratio of the transverse strain to the corresponding axial strain. for solving the Poisson-Boltzmann equation only little changes in a script generated by. THE Poisson ratio of a solid characterizes its response to uniaxial stress. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
